Summary: Photons are the real thing. Yes, by carrying all the energy in neat packets, photons are, in fact, light.
What are photons, which appear to the real thing that gives us the energy from the sun?
Put simply, one can say that “photons are light”. Yes, photons are pretty much what make light, and can hence be simplistically defined as the smallest particles of light.
Photons are super small packets of energy.
When we think of particles, we think of physical objects. That’s natural. But photons are funny. They are particles all right, but they also behave as waves.
Can you imagine a particle as a wave as well? In a way, you can: Visualise a wave in the ocean. The wave is comprised of water particles. Of course, this is a crude way of saying it and it does not really define light or photons, because in an ocean wave, there is a particle (water) that can be separated from the wave form – that is, I can take those water particles and make them move in other ways as well. But in the case of a photon, the particle and wave natures are intertwined in it and cannot be separated.
Not easy to visualize, right? This is why it required a certain Albert Einstein to figure these out, for which the nice bloke received the Nobel Prize.
When energy from photons is transferred to other particles, interesting things can happen. For example, if the photons hit an object and its energy makes the atoms and molecules in the object move faster, the object might become hot, or in some cases, even change shape.
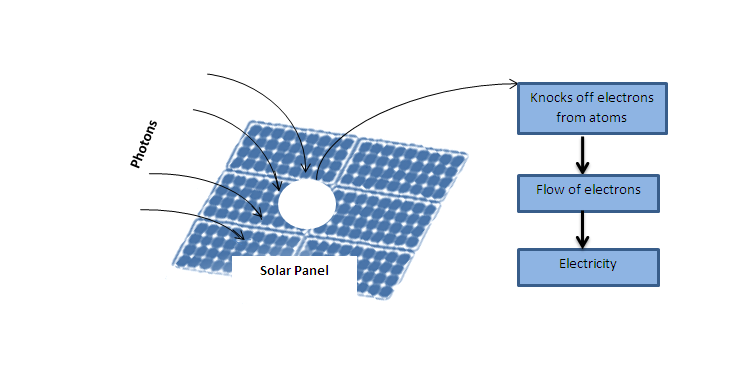
Of course, the other really interesting transfer of energy from photons happens in the case of solar power generation. Photons are the core of solar power. Through the photoelectric effect, the energy of light photons bump into the atoms of a semiconductor material. This causes the atoms to lose electrons. These electrons, when loosened, flow, and this constitutes electricity.
What an amazing entity, these photons are!

Questions from the curious cat – and answers
- How are these phenomenal photons created? I mean, from where do they originate, and how?
Nice that you have started at the very beginning. Isn’t that a nice question – how indeed are these powerhouses created?
OK, let’s keep this simple so you remember.
- Electrons revolve around a nucleus in any atom – you know this already
- Different electrons revolve in different orbits, some in orbits closer to the nucleus and some in orbits farther away
- In an atom, an electron far from a nucleus carries more energy than one that is close.
- When there is any event that makes an electron from a higher (farther) orbit fall into a lower (closer to nucleus) orbit, the difference in energy is released.
This energy gets nicely packaged as a photon.
- So, er, can we actually create protons ourselves?
Hmmm, why did we have a feeling this question was coming!
Can we create photons in real world? Of course, yes.
Sodium vapour lights are one instance where you can see the creation of photons. Along highways and in parking lots, you often see sodium vapor lights, those really bright yellow lights. A sodium vapor light energizes sodium atoms to generate photons. A sodium atom has 11 electrons, and because of the way they’re stacked in orbitals, one of those electrons from a higher orbital falls to a lower orbital, releasing energy as photons in the process.
There are many other such instances when these packets of energy are created, but the sodium vapor light is one of the most visible examples.
- How much energy does each photon carry?
Good question. The amount of energy of each photon depends on its frequency. And frequency is inversely related to wavelength, that is, higher the wavelength, lower the frequency of a photon. Photons that belong to those portions of light with lower wavelength (the gamma rays and the ultra-violet rays for instance) have much higher energy than those belonging to the infra-red rays (which have higher wavelength and hence lower frequencies)
- Can I see these awesome photons?
It is not surprising that many of you would like to see a picture of these phenomenal photons. But it appears that it is tremendously difficult, if not impossible, to see photons.
Well, look at it this way: In order to see anything, you have to first absorb the light from it. But what happens when you have to first absorb what you wish to see in order to see it?
“Hang on, this is getting a bit twisted”, you are saying. We hear you.
So, I repeat: In order to see anything, your eyes have to first absorb the light from it. But what happens when you have to first absorb the very photons before you can see them?
In a way, it is almost similar having to “destroy” an object before you could see it, isn’t it? This is the case with the photon. At least with the current level of understanding and technology, it is not going to be easy for you to “see” a photon. Sorry about that.
- These phenomenal photons, how much does each weigh?
Funny as it sounds, photons do not have a mass. Yes, we describe them as particles, but they simply do not have any mass.
Sometimes, you have to take life easy and just accept things as they are. Period.
Some nice videos for you
The voyage of photons
A short video, but an eye-opener on these awesome photons
 Skip to content
Skip to content

Very interesting blog about photons. Specially the conclusion. We have to destroy the object in order to see that object.. Pretty interesting..